A new collaboration between two UC Santa Barbara labs explores the underlying molecular mechanism of a remarkable process called anastasis, a Greek word meaning “rising to life.” Building on earlier work showing that cells can return from the brink of death, the new study demonstrates that anastasis is an active process composed of two distinguishable stages. The team’s findings appear in the Journal of Cell Biology.
“We knew already that cells need to transcribe new genes in order to recover,” explains corresponding author Denise Montell, UCSB’s Duggan Professor of Molecular, Cellular, and Developmental Biology. “So we profiled every molecule of mRNA in the cells as they started to die and then as they recovered.”
First, the biologists added a toxin to the growth medium to induce apoptosis — a form of programmed cell suicide that is an integral part of almost every disease — and took the cells to the brink of death. They then exchanged the medium to remove the inducer and allowed the cells to recover for one, two, three, four, eight or twelve hours. At every step, the researchers collected millions of cells and sequenced their RNAs to discover how their genetic profile changed during this process. UCSB’s Kosik Molecular and Cellular Neurobiology Lab conducted the RNA analysis.
The data from the RNA profiles not only demonstrated the active nature of the anastasis process, but also showed its two distinct phases. Patterns of gene expression resembled each other at the one-, two-, three-, and four-hour intervals, as did those at eight and twelve hours of recovery. Both groups were much different from untreated cells.
“We also found that even when cells are at the brink of death, they are secretly enriching survival RNAs,” Montell said. “The cells don’t know if things are going to get better or worse, so they hold on to some survival molecules just in case. The cells are poised to recover even while they’re dying.”
The team focused on one particular pro-survival RNA, called “snail,” which is enriched at the brink of death. The cells don’t make protein out of the RNA or degrade it; rather, they hold on to it. When the scientists prevented the expression of snail, the cells were unable to survive.
They also discovered that RNAs induced in the early phase of anastasis promote transcription of other genes, which allows cells to recover and start dividing. In the later phase, RNAs change what they make and acquire the ability to migrate.
“Some things are expressed during the whole recovery process, including angiogenesis inducers that make new blood vessels,” Montell notes. “This looks a lot like wound healing — cell proliferation or migration to fill in the gap and the creation of new blood vessels to nourish the recovery.
“That’s all fine in a normal beneficial process,” she adds. “For example, it's good news that cells deprived of oxygen during a heart attack can recover. But when cancer cells do the same thing, it’s bad news. Chemotherapy drugs and radiation are known to induce cancer cells to undergo apoptosis. But anastasis may give them a way to bounce back after treatment.”
Now that the researchers have described this molecular mechanism, they are particularly interested in the earliest phases of recovery before cells begin transcribing new genes. They also would like to better understand the long-term cellular effects of anastasis. “We want to know whether a cell recovering from the brink of death retains a permanent epigenetic memory of the experience,” Montell said. “We also want to find out whether cells that have experienced one round of anastasis are more or less resilient to a subsequent round. And most importantly, does the mechanism we describe in this paper underlie relapse after chemo and radiation therapy?”
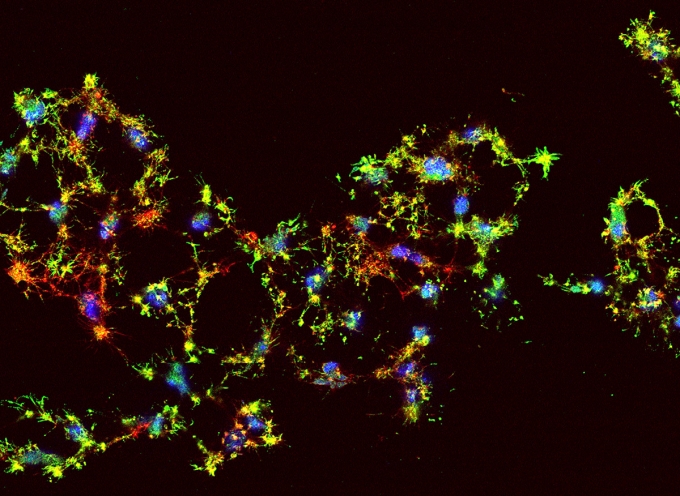
Dying human cancer cells are labeled with flourescent dyes to show DNA (blue), actin (red), and active caspase 3 (green).
